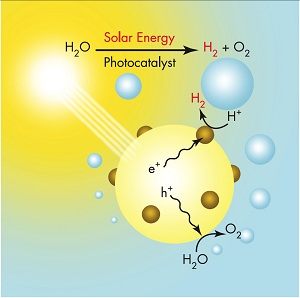
 The hydrogen economy is a worthy dream, given that it could provide an abundant source of non-polluting renewable energy. There are some roadblocks on the way to it, though. One is that it’s much cheaper to produce hydrogen from gas than it is from water. Gas is a fossil fuel, it is not renewable, and the process is not non-polluting. That’s fine for now because gas is cheap and plentiful, thanks to fracking, and the pollution is far less than from coal. The hope is though, that we can produce our hydrogen in future by splitting water, which is not a fossil fuel, is in abundant supply, and can be done in a non-polluting way. The trick might be to use solar cells to generate electricity, and use that electricity to split water. But it has to become more efficient. The problem is that the silicon in the solar cell that produces the oxygen half of electrolysis corrodes within hours. Now MIT Technology Review reports that Stanford scientists have found that by coating the silicon with a layer of zinc only two-billionth of a metre thick, they can make it last for days, and possibly much longer. Furthermore, the process is an order of magnitude faster than that achieved by other durable coatings such as metal oxides. The holy grail is not here yet, but it might just have moved close enough for us to make out its outline. Efficient and durable solar cells combine electricity production with electrolysis to split water, and we use the hydrogen to power our engines while emitting only water. Until then there’s plentiful gas and solar electricity is steadily becoming cheaper. It looks very much as if our energy needs can be met for at least a century, after which there’ll no doubt be new ideas developed.
The hydrogen economy is a worthy dream, given that it could provide an abundant source of non-polluting renewable energy. There are some roadblocks on the way to it, though. One is that it’s much cheaper to produce hydrogen from gas than it is from water. Gas is a fossil fuel, it is not renewable, and the process is not non-polluting. That’s fine for now because gas is cheap and plentiful, thanks to fracking, and the pollution is far less than from coal. The hope is though, that we can produce our hydrogen in future by splitting water, which is not a fossil fuel, is in abundant supply, and can be done in a non-polluting way. The trick might be to use solar cells to generate electricity, and use that electricity to split water. But it has to become more efficient. The problem is that the silicon in the solar cell that produces the oxygen half of electrolysis corrodes within hours. Now MIT Technology Review reports that Stanford scientists have found that by coating the silicon with a layer of zinc only two-billionth of a metre thick, they can make it last for days, and possibly much longer. Furthermore, the process is an order of magnitude faster than that achieved by other durable coatings such as metal oxides. The holy grail is not here yet, but it might just have moved close enough for us to make out its outline. Efficient and durable solar cells combine electricity production with electrolysis to split water, and we use the hydrogen to power our engines while emitting only water. Until then there’s plentiful gas and solar electricity is steadily becoming cheaper. It looks very much as if our energy needs can be met for at least a century, after which there’ll no doubt be new ideas developed.
Filed under: Updates |









We are often confronted with the question of suitable means of storing electrical energy. Large scale storage using any known form of rechargeable batteries is a relative no-no because the scale involved would be prohibitive. However, converting electrical energy into other forms of usable energy is wide open to research and innovation. Mega-solar cell installations could provide hydrogen generation by large scale electrolysis of water. The conversion is inefficient but the energy source to carry this out is free forever. When the cost of solar cells reduce to a point of financial viability this process could perhaps be achieved. Is hydrogen powered technology our future ? I seem to recall reading some time ago that the oceans of the world could provide enough hydrogen to meet mankind’s requirements forever.